Seznamy Draw An Atom Of Sodium
Seznamy Draw An Atom Of Sodium. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. The rings represents electron shells and the x represents the electrons.
Nejchladnější 3 2 Ions The Basics Of General Organic And Biological Chemistry
Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring. The first valence shell contains 2 electrons, and the … Views what does the bohr diagram for aluminum look like?.04.01.2015 · we draw atomic structures for any element with the help of atomic number they have.
So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. Views what does the bohr diagram for aluminum look like?. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. The na which is the word at the center of the ring is the atom/element. Atomic number of any element refers to the number of electrons in an atom of that element they are having.

Sodium atom electronic configuration is 2.8.1.. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. The electronic configuration of sodium is: The na which is the word at the center of the ring is the atom/element. Views what does the bohr diagram for aluminum look like?. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. Atomic number of sodium is 11. The na which is the word at the center of the ring is the atom/element.

The number represent the number of electron on that ring.. Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells.. The na which is the word at the center of the ring is the atom/element.

In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. 13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? Always write the atomic number of the element first. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. Atomic number of any element refers to the number of electrons in an atom of that element they are having. Sodium atom electronic configuration is 2.8.1... Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x.

Sodium atom electronic configuration is 2.8.1. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. The sodium atom is drawn this way as its electronic configuration is (2.8.1). So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. 13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. Easier explanation is by following the electronic configuration. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring.. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons.

03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x. Easier explanation is by following the electronic configuration. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. Atomic number of sodium is 11.. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium.

Easier explanation is by following the electronic configuration. The rings represents electron shells and the x represents the electrons. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. The valence shell of the sodium atom is \3\.. Sodium atom electronic configuration is 2.8.1.

So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. Sodium atom electronic configuration is 2.8.1. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\.

Easier explanation is by following the electronic configuration. The first number represent the first ring, second number is the second ring. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. The rings represents electron shells and the x represents the electrons. Easier explanation is by following the electronic configuration. Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\. It has a total of \11\ protons and \11\ electrons.

The first valence shell contains 2 electrons, and the ….. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. The na which is the word at the center of the ring is the atom/element. The number represent the number of electron on that ring. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. Atomic number of any element refers to the number of electrons in an atom of that element they are having. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\. Steps to draw the bohr model of sodium atom 1.. Atomic number of any element refers to the number of electrons in an atom of that element they are having.

13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium?. 03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x.

The first valence shell contains 2 electrons, and the …. The sodium atom is drawn this way as its electronic configuration is (2.8.1). Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. The electronic configuration of sodium is: Atomic number of sodium is 11. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. Steps to draw the bohr model of sodium atom 1. The number represent the number of electron on that ring.. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles.

It has a total of \11\ protons and \11\ electrons. The valence shell of the sodium atom is \3\. The first number represent the first ring, second number is the second ring. Always write the atomic number of the element first. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons.

04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells.. Easier explanation is by following the electronic configuration.

So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. Steps to draw the bohr model of sodium atom 1. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. The first valence shell contains 2 electrons, and the … Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring.

Steps to draw the bohr model of sodium atom 1. Atomic number of sodium is 11. Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\. Atomic number of any element refers to the number of electrons in an atom of that element they are having. The first valence shell contains 2 electrons, and the … In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively.. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively.

The sodium atom is drawn this way as its electronic configuration is (2.8.1). Sodium atom electronic configuration is 2.8.1. Atomic number of any element refers to the number of electrons in an atom of that element they are having. It has a total of \11\ protons and \11\ electrons. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring.

Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\. Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. 03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x. It has a total of \11\ protons and \11\ electrons. Atomic number of any element refers to the number of electrons in an atom of that element they are having.

In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively.. Easier explanation is by following the electronic configuration... Sodium atom electronic configuration is 2.8.1.

Atomic number of any element refers to the number of electrons in an atom of that element they are having. The rings represents electron shells and the x represents the electrons. It has a total of \11\ protons and \11\ electrons. Sodium atom electronic configuration is 2.8.1.. Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\.
Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring.. Atomic number of sodium is 11. The rings represents electron shells and the x represents the electrons. Steps to draw the bohr model of sodium atom 1. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring... The number represent the number of electron on that ring.

13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? The rings represents electron shells and the x represents the electrons... Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells.

03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x. 13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? Atomic number of any element refers to the number of electrons in an atom of that element they are having. Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. The rings represents electron shells and the x represents the electrons. It has a total of \11\ protons and \11\ electrons. Easier explanation is by following the electronic configuration. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. The na which is the word at the center of the ring is the atom/element... The first number represent the first ring, second number is the second ring.

So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. 13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? The sodium atom is drawn this way as its electronic configuration is (2.8.1). Always write the atomic number of the element first. The na which is the word at the center of the ring is the atom/element. Sodium atom electronic configuration is 2.8.1. The first number represent the first ring, second number is the second ring. Atomic number of sodium is 11. The sodium atom is drawn this way as its electronic configuration is (2.8.1).

13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? The valence shell of the sodium atom is \3\. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. It has a total of \11\ protons and \11\ electrons. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. The number represent the number of electron on that ring. The rings represents electron shells and the x represents the electrons. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. Always write the atomic number of the element first.

Always write the atomic number of the element first.. The sodium atom is drawn this way as its electronic configuration is (2.8.1). So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. The na which is the word at the center of the ring is the atom/element.
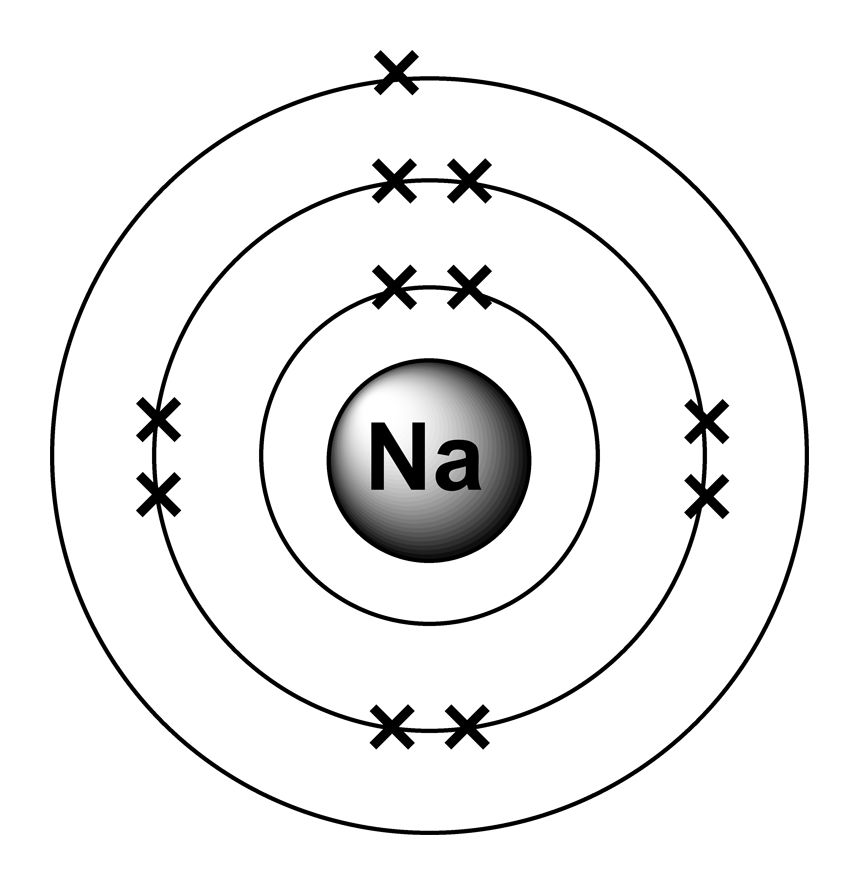
Steps to draw the bohr model of sodium atom 1. Steps to draw the bohr model of sodium atom 1. The sodium atom is drawn this way as its electronic configuration is (2.8.1). Atomic number of sodium is 11. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. Always write the atomic number of the element first. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure.. Easier explanation is by following the electronic configuration.

The sodium atom is drawn this way as its electronic configuration is (2.8.1)... Atomic number of any element refers to the number of electrons in an atom of that element they are having. Steps to draw the bohr model of sodium atom 1.

Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. The valence shell of the sodium atom is \3\. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles.. Easier explanation is by following the electronic configuration.

Sodium atom electronic configuration is 2.8.1.. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. The valence shell of the sodium atom is \3\. So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. Atomic number of any element refers to the number of electrons in an atom of that element they are having. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. The number represent the number of electron on that ring. The na which is the word at the center of the ring is the atom/element. Sodium atom electronic configuration is 2.8.1. The first valence shell contains 2 electrons, and the ….. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure.

A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. Steps to draw the bohr model of sodium atom 1. 03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x. Always write the atomic number of the element first.

Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. Views what does the bohr diagram for aluminum look like?. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. The valence shell of the sodium atom is \3\. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. The sodium atom is drawn this way as its electronic configuration is (2.8.1). Sodium atom electronic configuration is 2.8.1. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring... Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of.

Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. Steps to draw the bohr model of sodium atom 1. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x... Atomic number of sodium is 11.

The valence shell of the sodium atom is \3\. The rings represents electron shells and the x represents the electrons. Atomic number of any element refers to the number of electrons in an atom of that element they are having. The first number represent the first ring, second number is the second ring. Views what does the bohr diagram for aluminum look like?. The number represent the number of electron on that ring. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. 13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring. The sodium atom is drawn this way as its electronic configuration is (2.8.1). Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge.

The electronic configuration of sodium is:.. It has a total of \11\ protons and \11\ electrons.
Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons... . In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively.

Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. . Easier explanation is by following the electronic configuration.

Easier explanation is by following the electronic configuration.. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. It has a total of \11\ protons and \11\ electrons. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. The rings represents electron shells and the x represents the electrons. 13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? The electronic configuration of sodium is: Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles... The na which is the word at the center of the ring is the atom/element.

Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. 03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x. Atomic number of sodium is 11. The number represent the number of electron on that ring. The rings represents electron shells and the x represents the electrons.. Always write the atomic number of the element first.
Steps to draw the bohr model of sodium atom 1.. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. The na which is the word at the center of the ring is the atom/element. Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. The rings represents electron shells and the x represents the electrons.. Easier explanation is by following the electronic configuration.

Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons... 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have.. Steps to draw the bohr model of sodium atom 1.

13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? The first valence shell contains 2 electrons, and the …. Atomic number of sodium is 11.

The sodium atom is drawn this way as its electronic configuration is (2.8.1). Easier explanation is by following the electronic configuration. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. The first number represent the first ring, second number is the second ring. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. The electronic configuration of sodium is:. Steps to draw the bohr model of sodium atom 1.

Steps to draw the bohr model of sodium atom 1.. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. The rings represents electron shells and the x represents the electrons. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. The sodium atom is drawn this way as its electronic configuration is (2.8.1). It has a total of \11\ protons and \11\ electrons. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring. The first valence shell contains 2 electrons, and the … The na which is the word at the center of the ring is the atom/element.. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of.

Always write the atomic number of the element first. Sodium ion have 10 electrons, thus we have to draw 2 rings and 10 x. The first number represent the first ring, second number is the second ring. The na which is the word at the center of the ring is the atom/element. The rings represents electron shells and the x represents the electrons. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. Atomic number of any element refers to the number of electrons in an atom of that element they are having. Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have.. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge.

Neutral sodium atom has 11 electrons whereas sodium ion (n a +) has 10 electrons. The sodium atom is drawn this way as its electronic configuration is (2.8.1). The rings represents electron shells and the x represents the electrons. A sodium ion (na+) is essentially a sodium atom which had been oxidized, losing one electron and gaining a positive charge. Always write the atomic number of the element first. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. The first valence shell contains 2 electrons, and the … The na which is the word at the center of the ring is the atom/element. Steps to draw the bohr model of sodium atom 1. Sodium atom have 11 electrons ,thus we have to draw 3 rings around the wordna and the x on each of the ring.

Sodium atom electronic configuration is 2.8.1. Atomic number of sodium is 11. The na which is the word at the center of the ring is the atom/element. Sodium is an atom in the periodic table with atomic number \11\ and mass number\23\. The electronic configuration of sodium is: 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. The sodium atom is drawn this way as its electronic configuration is (2.8.1). Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. The first number represent the first ring, second number is the second ring. The rings represents electron shells and the x represents the electrons. Easier explanation is by following the electronic configuration.

Steps to draw the bohr model of sodium atom 1. Sodium atom electronic configuration is 2.8.1. 03.07.2011 · sodium atom have 11 electrons, which means we have to draw 3 rings and 11 x.

Easier explanation is by following the electronic configuration. Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles.. The valence shell of the sodium atom is \3\.

13.11.2018 · bohr model · diagrams how do you draw a bohr diagram for calcium? Find the number of protons, electrons, and neutrons in the sodium atom protons are the positively charged particles. The number represent the number of electron on that ring. 04.01.2015 · we draw atomic structures for any element with the help of atomic number they have. Atomic number of any element refers to the number of electrons in an atom of that element they are having.

Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. . The na which is the word at the center of the ring is the atom/element.

The na which is the word at the center of the ring is the atom/element. Draw the nucleus of an atom a nucleus is a dense and small region that contains the number of protons and neutrons of. Atomic number of any element refers to the number of electrons in an atom of that element they are having. So sodium chloride (and any other ionic compound) is described as having a giant ionic structure. Theoretically, they.a bohr diagram depicts an atom with a small, central nucleus and the electrons in their valence shells. The rings represents electron shells and the x represents the electrons. The number represent the number of electron on that ring. Sodium atom electronic configuration is 2.8.1.. Atomic number of any element refers to the number of electrons in an atom of that element they are having.

In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively. The valence shell of the sodium atom is \3\. The first number represent the first ring, second number is the second ring. Steps to draw the bohr model of sodium atom 1. In contrast, chlorine and sodium have seven and one electrons in their outer shells, respectively.
